Covid-19
Full authorization of Pfizer vaccine to be given by Monday, sources say
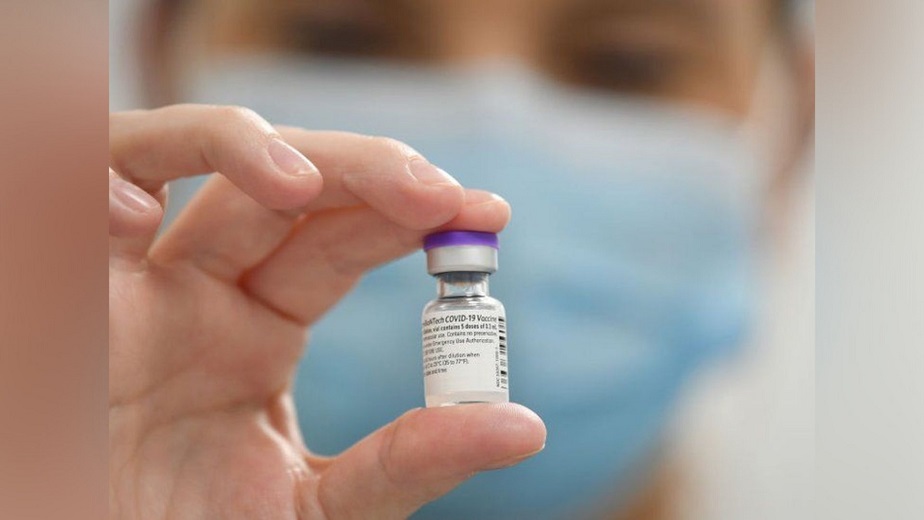
According to New York Times sources, the Food and Drug Administration is trying to give full authorization to Pfizer-BioNTech’s two-dose Covid-19 vaccine.
Authorities were attempting to complete the approval by Friday, but were still working on paperwork and negotiating with the company, according to the New York Times sources. They further claim that if more issues arise, the date might be pushed out even more.
The Food and Drug Administration had recently set a Labor Day deadline for the vaccine’s approval.
The agency updated its emergency authorizations for both Pfizer and Moderna last week to allow for third doses for some immunocompromised people.
A request for comment was denied by the Food and Drug Administration.





Leave a Reply
You must be logged in to post a comment.